SYNCRETIC has always given importance to quality, integrity and adherence to promised timelines. Following are our prime services.

Cost-effective studies are always welcome. SYNCRETIC ensures that the study budget is well handled and the study is completed within timelines hence saving a lot of miscellaneous costs to the Sponsor. Our effective team ensures that best solutions are provided at modest and competitive costs.

The Protocol design is as important as the drafting of the final Clinical Study Report. SYNCRETIC has a highly experienced medical writers who use their expertise to design a study in the best way possible so that the Sponsor is able to get the precise data that is expected out of a particular clinical research study.

The final output of any clinical study is dependent on expert statistical analysis, SYNCRETIC offers just that! Our statisticians have a decade’s experience in performing the statistical analysis for various clinical studies including but not limited to global, multicentric, parallel group, cross-over, placebo-controlled, standard-controlled, and the like.

The effective interpretation of the statistics is seen in the quality of the final clinical study report generated. SYNCRETIC has some of the best medical minds in drafting the report. This is the report that will be submitted by the Sponsor to the regulatory authorities and hence we ensure that it adheres to the regulatory standards.

Most Sponsor companies are on the lookout for journal publication support and not many CROs support the Sponsor post the submission of the Clinical Study Report. However, SYNCRETIC is few of those CROs that stand apart from the industry crowd. We do have an excellent support team that specializes in journal publications and ever ready to support the Study Sponsors.

Quality clinical data is the final outcome of any study. SYNCRETIC understands this aspect to the hilt. We ensure that the data generated is as per the Protocol requirements adhering to the local regulations and compliant with ICH-GCP (R2). This is our primary commitment to all our Sponsors.
SYNCRETIC has always given importance to quality, integrity and adherence to promised timelines. Contact us to know more about our services.


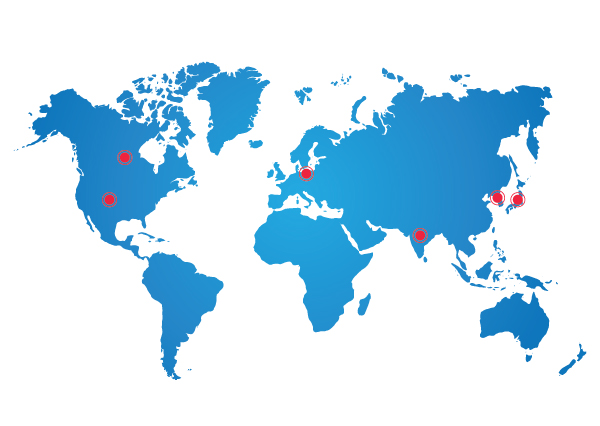
Syncretic is a global clinical research organization that specializes in providing exceptional clinical research services to its international clients across the world.
Syncretic offers you a one-stop solution for all your clinical research needs. Get in touch to know more.
A small step in clinical research can become a giant leap in global healthcare.